THE RISK MANAGEMENT OF THE PHARMACY PREPARATIONS IN THE HOSPITAL PHARMACIES (submitted in 2019)
Pdf

European Statement
Production and Compounding
Author(s)
ADRIANA DURCANSKA
Why was it done?
The quality and safety standards of pharmacy preparations are not harmonised throughout Europe. They fall under the national competencies of individual European countries.
What was done?
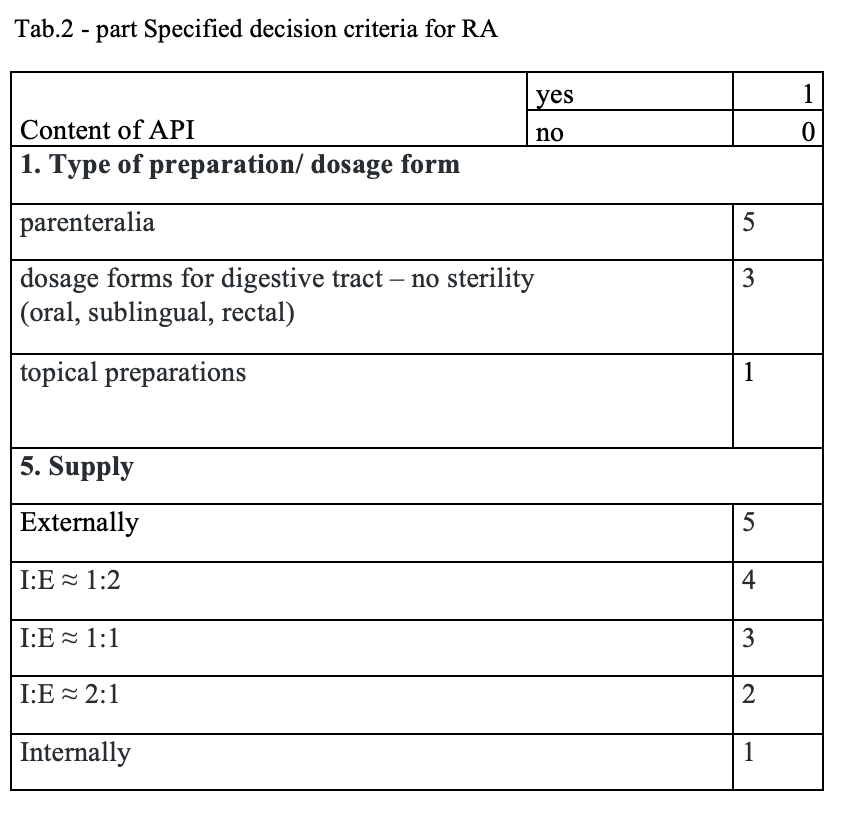
The quantitative risk assessment of the pharmacy preparations for stock in hospital pharmacies (HPs) in accordance with Resolution EDQM CM / Res (2016) 1; to specify the decision criteria for the risk assessment; the risk management of the pharmacy preparations for stock in the country; to design a check list of the risk assessment for extempore preparations.
How was it done?
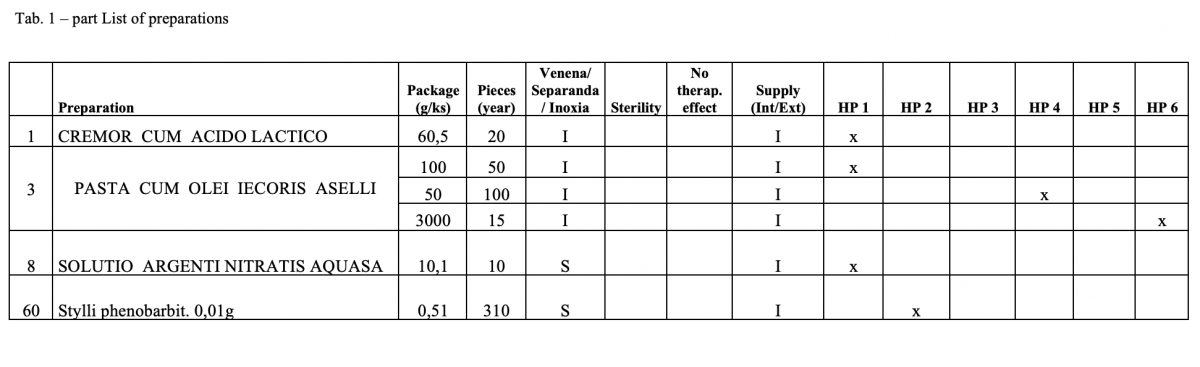
Out of the total number of 53 hospital pharmacies contacted, 5 pharmacies sent a suitable file.
What has been achieved?
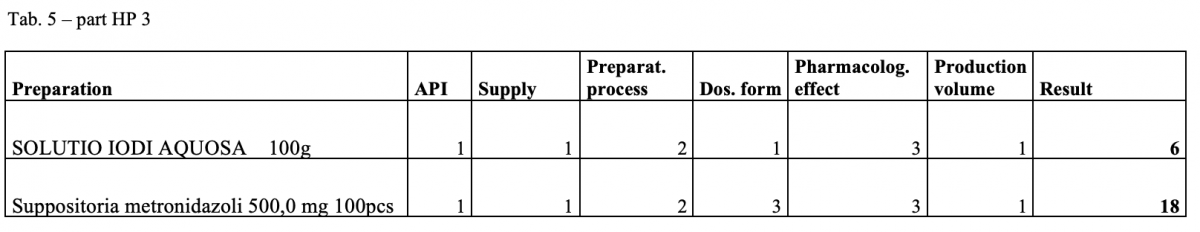
A total of 170 types of medicines are being prepared in HPs. One HP had the result of the risk ≥ 100 when preparing ophthalmic medicines. Annex A is a check list designed to assess the risk of extempore preparations.
What next?
The management is and will be forced to consider its introduction or to use another model: hospital – GMP / outsourcing / central pharmacy preparing and distributing. The aim of using the document in hospital pharmacies of the country.
TEMPERATURE AND RELATIVE HUMIDITY CONTROL IN THE PACKAGING ENCLOSURE OF SOLID ORAL DOSAGE FORMS (submitted in 2019)
Pdf

European Statement
Production and Compounding
Author(s)
María Lourdes Recio Blázquez, José Manuel Martínez Sesmero, Lidia Ybáñez García, Gonzalo Hernando Llorente, María Molinero Muñoz
Why was it done?
The purpose is to control two environmental conditions to guarantee the comfort of the workforce and the quality of the finished product quality. 519,321 SODFs have been repackaged in unit doses last year. The 8% of the SODFs come from multidose containers that have been exposed to environmental temperature and humidity during this process. Employees have been exposed to identical conditions.
What was done?
A temperature (t) and relative humidity (RH) control system has been established in the enclosure where the solid oral dosage forms (SODFs) are packaged in unit doses.
How was it done?
Among the diversity of hygrometric sensors commercialised, a device equipped with a condenser was chosen. The operation is based on modifying the capacity when varying the dielectric constant of the medium, in this case, due to varying the amount of water contained in the air between the plates. – C = ε A / D – C: capacity value. – ε: dielectric constant. – A: area of the condenser plates. – D: distance between the condenser plates. The device also incorporates a temperature sensor. The t (ºC) and RH of each moment are shown, for visual inspection, on the device screen. The data obtained with certain time frequency can be stored on a Secure Digital memory card and be downloaded on a computer that has that program installed (on spreadsheet format) helping to obtain graphics as well.
What has been achieved?
The range of t (ºC) has remained stable between 26 and 24ºC for 6 months, with minimal variations from maximum 28.5ºC to minimum 23.4ºC. The UNE 100713: 2005 is met. RH has been below 45% during 68% of the days worked, which has favored the repackaging of the units affected by humidity but not the worker. The range of RH has varied between 56.3% and 23.6%, not complying with the UNE 100713: 2005 standard.
What next?
Metabolic rate, clothing insulation, air temperature, radiant temperature, air speed and humidity shall be addressed when defining conditions for acceptable thermal comfort. It would be helpful to regulate the commercialisation of multidose pharmaceutical specialties susceptible to deterioration when opening the package.
INTEGRATION OF A ROBOT INTO THE EXISTING WORKFLOW OF THE CYTOSTATIC DRUGS DEPARTMENT IN A HOSPITAL PHARMACY (submitted in 2019)
Pdf

European Statement
Production and Compounding
Author(s)
Swantje Eisend, Herwig Heindl, Karen Tiede, Sven Jirschitzka
Why was it done?
Definition of an organisational structure for the best implementation of APOTECAchemo technology in the UKSH hospital pharmacy workflow
What was done?
The implementation of robotic systems for aseptic compounding cytotoxic drugs requires a specific workflow organisation in the hospital pharmacy to ensure an optimal combination of manual and automated production as well as the effective use of the technology. Since 2017, the APOTECAchemo robot has been installed in the hospital pharmacy and one of the first objectives was to create an organisational structure that would allow successful integration of the system into the existing workflow of the cytostatic department.
How was it done?
The pharmacy has carried out an analysis to identify the active substances that can best be transferred into automated production based on 4 main points: • Pharmaceutical form of the active ingredients: liquid or powder; • Average of vials needed for the compounding of one preparation for each active ingredient; • Average of ml of medication required for the compounding of a preparation associated with each specific active ingredient; • Robot compounding speed. In addition, the pharmacy has also tried to identify the optimal organisation of personnel and daily workflow for the automated compounding. The effectiveness of these measures and the work organisation defined have been evaluated through an intensive compounding week in April 2018.
What has been achieved?
The analysis of the active substances and the data collected during the “Robotic Intensive Week” showed the following results: • 42% of the total production was operated by APOTECAchemo; • 87% of active ingredients was handled by APOTECAchemo; • average of 60 preparations per day (with an actual working time of 5 hours); – average of 12 preparations per hour.
What next?
The study shows that the planning and organisation of the workflow plays a central role in the implementation of a robot solution in a hospital pharmacy. Through the work carried out, the hospital pharmacy has successfully integrated automated and manual production.
HOW TO HANDLE ACTIVE AND PLACEBO DRUGS FOR A CLINICAL TRIAL IN THE PRODUCTION SYSTEM CATO IN ORDER TO SECURE THE BLIND AND TO ENSURE THE EXACT SAME PREPARATION OF ACTIVE AND PLACEBO PRODUCTS
Pdf

European Statement
Production and Compounding
Author(s)
Kirsten Lykke Vorbeck
Why was it done?
When preparing treatments for blinded clinical trials it is very important to make sure that it is not possible to distinguish between the active and the placebo dose, as this may lead to unblinding of the treatment and thus jeopardise the results of the trial.
What was done?
The purpose was to find solutions for how to handle active and placebo drugs in the production system CATO to ensure they would end up being identical when prepared.
How was it done?
Before preparing a drug using CATO the drug must be registered in the system with all of its data. It is not possible to have two different sets of data for one drug. CATO records detailed documentation of what is being done during preparation and it is not possible to pretend to pick one drug and then grab another instead. Also, the label is printed automatically so it will show exactly what was prepared. We decide on a common name for the active/placebo and register both of these in the system under that name so that they will be prepared in exactly the same way and appear identical on the label but are still distinguishable in the batch documentation. We name the drug ‘Protocol name Active substance/placebo’ and register the stock of both active drug and the sodium chloride solution that is used as placebo under that name. We will also stock vials of NaCl for this purpose next to the drug it is being placebo for, and NaCl can be prepared pretending it has the density of the drug. CATO will think they are the same drug but they have been registered with their very different batch numbers, maybe with a NaCl after the number of that, so it is always possible to see what was actually being prepared.
What has been achieved?
Our doses of active and placebo are indistinguishable. Labels are the same and, for example, withdrawal into a syringe and insertion of a needle into the port of the infusion bag will be done in the same way.
What next?
These very simple routines can be used anywhere for the preparation with CATO and possibly also with other systems.
DEVELOPMENT OF NEW PRODUCTION WHEN NEITHER PACKAGING NOR SOME OF THE RAW MATERIALS CONFORM TO EUROPEAN STANDARDS
Pdf

European Statement
Production and Compounding
Author(s)
Katrine Bødker Rubach-Larsen, Anne Rungø, Anette Eskildsen, Lone Skovhauge
Why was it done?
A research team at the MR Centre (MRC2) wished to set up the production of Pharmacy Kits, but had no prior experience of, or licence to, manufacture drugs. Thus, the hospital pharmacy was asked to participate in the development of such production.
What was done?
A new MR-scanning technology, hyperpolarisation, for the quantification of metabolic processes with an extremely high sensitivity enables physicians early detection of treatment effects in, for example, cancer and diabetes. A so-called Pharmacy Kit is used in the hyperpolarisation process and consists of a specially designed packaging with tubes, vessels and filters containing the contrast agent and buffer solutions. The objective for the hospital pharmacy1 was to manufacture Pharmacy Kits complying with Good Manufacturing Practice (GMP), though neither packaging nor two of the raw materials conformed to European standards.
How was it done?
The MRC research team presented the hospital pharmacy with the desired combination of compounds and the packaging required for Pharmacy Kit production. The task for the hospital pharmacy was then to set up a manufacturing process that met these requirements and complied with the guidelines for GMP. A production complying with GMP was developed in close collaboration with the MRC and an ongoing contact with the Danish Medicines Agency. During the process the hospital pharmacy carried out its own microbiology test in order to determine if, and for how long, the non-CE-marked packaging could store the contrast agent and buffer solutions. Risk assessment of the raw materials not found in the European Pharmacopeia were conducted. The method investigated by the MRC already takes place at a few other places in and outside of Europe. Experiences from these production sites were implemented and expanded with process optimisation, and specially designed equipment for the production.
What has been achieved?
Due to a strong inter-professional collaboration between the MRC and the hospital pharmacy and due to qualified risk assessments, it was possible to set up a production of Pharmacy Kits according to GMP.
What next?
When researchers contact hospital pharmacies with new ideas, we have to be willing to work with GMP in a different way by applying knowhow and risk assessments in order to ensure developments within the healthcare system.
1. Hospital Pharmacy Central Region, Production, Aarhus, Denmark.
2. MR Centre, Aarhus University Hospital, 8200 Aarhus N, Denmark
QUANTIFICATION OF WAITING TIME REDUCTION IN OUTPATIENT SETTING USING ASSISTED SYSTEMS IN AN AUTOMATED ONCOLOGY PHARMACY
Pdf

European Statement
Production and Compounding
Author(s)
Jemos Costantino, Martina Milani, Mariantonietta Piccoli, Mara Provenzi, Paola Paochi, Ilaria Clerici, Cinzia Lucia Ursini, Claudio Colosio, Fabrizio Mastrilli, Emanuela Omodeo Salè
Why was it done?
In outpatient setting, WT between medical visits and administration is strongly conditioned by time needed for preparation. We needed to reduce our WT caused by the use of an automated system by ensuring the same standards of quality control checks and traceability, not achievable with manual preparation.
What was done?
We introduced an assisted system for chemotherapy preparation, with a gravimetric and barcode verification. We started to switch part of preparations previously prepared by an automated system to this assisted system. We performed an analysis to measure the impact of a different strategy in preparing chemotherapy on patients WT.
How was it done?
Time needed for preparation was monitored in the first trimester of 2016, where drugs were prepared using an automated system or manually, and compared to the first trimester in 2017, when we introduced the assisted system.
In the first period we used a “WT optimization” criteria in selecting the preparing technology, while in the second period we decide to use a “risk based” criteria.
Risk based criteria consists of selecting the automated system for cytotoxics, assisted system for antibodies and low risk drugs and manual procedure when no other options are available.
In order to evaluate bias introduced by the risk based selection of different drugs, we performed a contest comparing preparation times of a defined sequence of representative preparations typologies. Three technicians are involved in order to reduce human factor impact.
What has been achieved?
Average WT (AWT) in the first period was 1h36m and median WT (MWT) was 1h22m (sample = 2365 preparations in 3 months). AWT in the second period (sample = 3437 preparations in 3 months) was 1h17m (-19,79%) and MWT 1h1m (-25,61%). The percentage of therapies dispensed after 2 hours waiting decreased by 55,69%. WT was stratified by preparation technology (assisted system: AWT =50m; MWT=44m – automated system: AWT=1h26m; MWT=1h07m).
The contest results were (average of three series) : manual preparation 15m19s; assisted 26m42s; automated 1h14m3s.
What next?
Assisted systems are able to guarantee quality standards for patients similar to automated ones, but with an important reduction in WT when compared with an automated one and an improvement in traceability compared to the manual procedure.
IMPLEMENTING NEW CHEMOTHERAPY COMPOUNDING WORKFLOW USING SEQUENTIAL PDSA CYCLES TO INCREASE PRODUCTION CAPACITY, REDUCE WAITING TIME, IMPROVE RESOURCES UTILIZATION, AND MINIMIZE THE RISK OF ERRORS
Pdf

European Statement
Production and Compounding
Author(s)
A Elsheashaey, A Elshishiny, A Orabi, A Almutairi, A Aboulwafa, H Alobaid, F Dashti, D Saeed, R Yassin, M Salama
Why was it done?
Kuwait Cancer Control Center (KCCC) is the only oncology hospital in Kuwait. Chemotherapy Preparation Unit (CTPU) was unable to meet the increased orders; causing delivery delay and more patients’ waiting time. Moreover; rework and more waste due to defective and faulty processes of current workflow resulting in frequent incident reports of wrong final products dispatched.
What was done?
Shifting to a systematic multi-step production workflow to increase compounding capacity, minimize risk of errors, reduce processing time, and maximize utilization of integrated technological resources.
How was it done?
Using multiple PDSA cycles, a comprehensive educational and practical training was conducted, proceeded by staff rotation with newly trained team. Every three weeks a new pharmacist trained and assigned to CTPU. Raw materials stores were rearranged for better accessibility and diminishing unnecessary staff movement. A staging step as the first independent double-check before the start of compounding, and for assembly of raw materials and supplies required for compounding. A verification Step as the second independent double-check upon compounding, using bar-code scanners, touchscreens and cross-checking with the chemotherapy order to assure the quality and integrity of the finished product. Production workload were restructured over three parallel line of manual stations and one automated preparation unit. Pharmacy Information System (PIS) screens were customized to give a first and second audio-visual alarms after 30 and 45 minutes of transcription time respectively. Chemotherapy sessions appointment system were established to assess the daily chemotherapy compounding needs from CTPU in advance with an incremental increase of production capacity to reach 100~120 patients/day or 180~200 preparations/day.
What has been achieved?
Number of preparation compounded by CTPU was increase by 8%, where more than 43% of preparations were validated to release in less than 30 minutes and approximately 88% of preparations were delivered in less than 45 minutes. Number of preparation by automation was increased by 82%, Furthermore, all production incidences has been completely eliminated after full implementation of final verification and validation step.
What next?
The new workflow has increase the workload capacity with less production errors and zero incident reports. Patient experience was improved by comparable preparation time to other international Pharmacy Workload Unit and average time required per patient visit.
IMPLEMENTATION OF INDIVIDUAL, HOSPITAL PHARMACY-COMPOUNDED NEONATAL TPN
Pdf

European Statement
Production and Compounding
Why was it done?
Individual total parenteral nutrition (TPN) for neonates was originally compounded by nursing staff on the respective wards. This process of TPN compounding was error-prone. Documentation and traceability was inadequate. Clean room conditions were absent. By transferring the compounding of TPN from the ward to the pharmacy level, several aims were accomplished. Time of nursing staff was released and the highest quality standards for compounding were implemented. By doing so, several types of errors (e.g. overdosing, wrong additives) were eliminated.
What was done?
Development and implementation of nutrition support protocols by using an electronic prescribing and compounding software (catoPAN™) to address the special needs of neonates and ensure a high level of individualized care.
How was it done?
In cooperation with neonatologists, nutrition protocols were developed. Furthermore, a TPN compounding process was implemented and validated, including the validation of catoPAN™ software and compounding pumps. An integrated risk analysis was performed, stability data to allow TPN supply for weekends were generated and fail-safe procedures were determined. To finally succeed, various process and organizational changes concerning the wards, the production and the QC department of the hospital pharmacy were required.
What has been achieved?
Compounding of individualized nutrition solutions within defined standards, predetermined specifications and quality attributes is implemented. The production process is continuously monitored, including complete traceability. A strong interprofessional collaboration between physicians, nurses and pharmacists was established, ultimately leading to a high level of confidence among all members. Workload of nurses in terms of compounding medicines was dramatically reduced.
Currently, we provide nutrition bags for four wards (24 ICU- and 30 intermediate care beds), equaling an average production of 50 bags per day. In 2016, a total of 11.126 bags were supplied, implying an increase of 75% compared to 2015. We expect an increase of around 30% in 2017 due to rising demand.
What next?
With the expansion of TPN compounding to further pediatric wards, new nutrition protocols addressing other requirements have to be developed. Process changes are likely to follow. Further support can be provided by pharmacy-based IV admixture service. Additionally, due to current software updates, the prescribing and compounding software catoPAN™ must continually be revalidated.
COMPUTERIZED QUALITY CONTROL OF THE TABLET SPLITTING PROCESS
Pdf

European Statement
Production and Compounding
Author(s)
María Lourdes Recio Blázquez, Alberto Pérez Morales
Why was it done?
It was necessary to establish a quality control of this pharmaceutical process.
What was done?
A computer method of gravimetric quality control of the tablet splitting process was designed.
How was it done?
The procedure consists on a precision scale connected to a computer in which, according to the uniformity of mass assay of the European Pharmacopoeia, the weights of 20% of a batch of whole tablets destined to be split are automatically recorded in a spreadsheet, carrying out the following formulas:
=AVERAGE: provides the average weight of the sample of whole tablets.
=MAX and =MIN: selects respectively the largest and the smallest of the weights.
=STDEV: calculates the standard deviation of the sample weights.
With the average weight of the whole tablets, the theoretical weight of the half-tablets is calculated, establishing a maximum and a minimum admissible limit with the following formulas:
=AVERAGE(whole tablets)/2: determines the theoretical average weight of each half-tablet.
=AVERAGE(whole tablets)/2 ± 7.5%: establishes upper and lower gravimetric limits that cannot be exceeded by any half-tablet.
All the half-tablets need to be weighted, as the tablet-splitting process is carried out tablet-by-tablet and this modus operandi is not reproducible enough. In case of non-compliance with maximum and minimum weight criteria, the half-tablet must be discarded.
Conditional functions were established such that the spreadsheet itself reflects the half-tablet acceptance/rejection decision.
Basic technical computer skills, training in the technique of tablet splitting, appropriate clothing and environmental measures to avoid risks to the operator and the medications are required.
What has been achieved?
Since 2015, two different medicinal products were subjected to the tablet splitting technique. A total of 10,536 halves of suitable tablets were obtained, which permitted safe dosing at lower doses than commercialized, and also generated a financial asset of 101,724 Euros. 566 halves were discarded. The splitting efficiency was of 94.9%.
What next?
This quality control procedure is applicable to all divisible solid oral dosage forms. The standardization of the technique and the quality controls will allow to extend it to other medicinal products with dosing and economic purposes.
DEVELOPMENT OF A COMPUTER APPLICATION TO REDUCE THE RISK OF ERRORS IN RECONSTITUTION OF CYTOTOXIC DRUGS
Pdf

European Statement
Production and Compounding
Why was it done?
The preparation of cytotoxic drugs perfusions can be accompanied by errors that can be fatal to some patients. Several methods were tested to minimise the risk of errors associated with reconstitution of cytotoxic perfusions (camera, HPLC, analytical balance, the presence of a second technician to monitor his colleague). All of these methods can be expensive and are not available to all hospital pharmacists.
What was done?
We present a simple and effective method that we have developed in our hospital to solve the problem of reconstitution errors.
How was it done?
Errors of cytotoxic drugs reconstitution can have catastrophic consequences for patients. Some studies have found that the incidence of major and minor errors were, respectively, 0.19% and 0.26%. Reconstitution control methods are numerous but not always accessible to all hospital pharmacists and preparers, particularly in developing countries.
This work involves the development of a computer application developed from an Open Source voice recognition software. The daily chemotherapy protocols are entered in the application that dictates to a technician the protocol prescribed by the physician, product by product, for all patients. The technician performs the cytotoxic preparations in the order in the centralised pharmaceutical unit for the preparation of cytotoxic drugs and he communicates with the application manually or by speaking.
The application begins with the patient’s ID, name and surname, the first drug, the dosage, the dilution solution and the volume of this solution. Then, it passes to the second product and so on. In the case of an observation, the application warns the technician to take this observation into account.
What has been achieved?
This application has helped provide better assistance to the technicians and pharmacists in the reconstitution of cytotoxic drugs, and no event or error has been detected to date.
What next?
To make a large number of reconstructions using this application to assess its effectiveness and install it in other hospitals who handle cytotoxic drugs.


























